Have you ever wondered why a wilted piece of celery perks up after being soaked in a liquid, or why a wilted piece of celery perks up after being soaked in water? These everyday mysteries are all driven by a fascinating biological process called osmosis. Bringing this high-level concept down to earth doesn’t require a high-tech laboratory; in fact, you likely have everything you need in your pantry right now. The potato osmosis experiment is one of the most effective, hands-on ways to show children how cells interact with their environment. By using a humble vegetable, you can transform your kitchen table into a discovery zone where the invisible movement of moisture becomes a tangible, measurable reality.
This science project is a staple in classrooms worldwide, from primary school to GCSE biology, because it provides clear, visual results. Whether you are a parent looking for a weekend simple science activity or a teacher seeking a reliable potato lab to demonstrate the concept of osmosis, this guide covers it all. This guide explores how water moves across a semi-permeable membrane, why concentration matters, and how to compare the results like a real scientist. Get ready to watch your pieces of potato shrink, swell, and teach your kids about the building blocks of life!
Potato Osmosis Experiment Overview
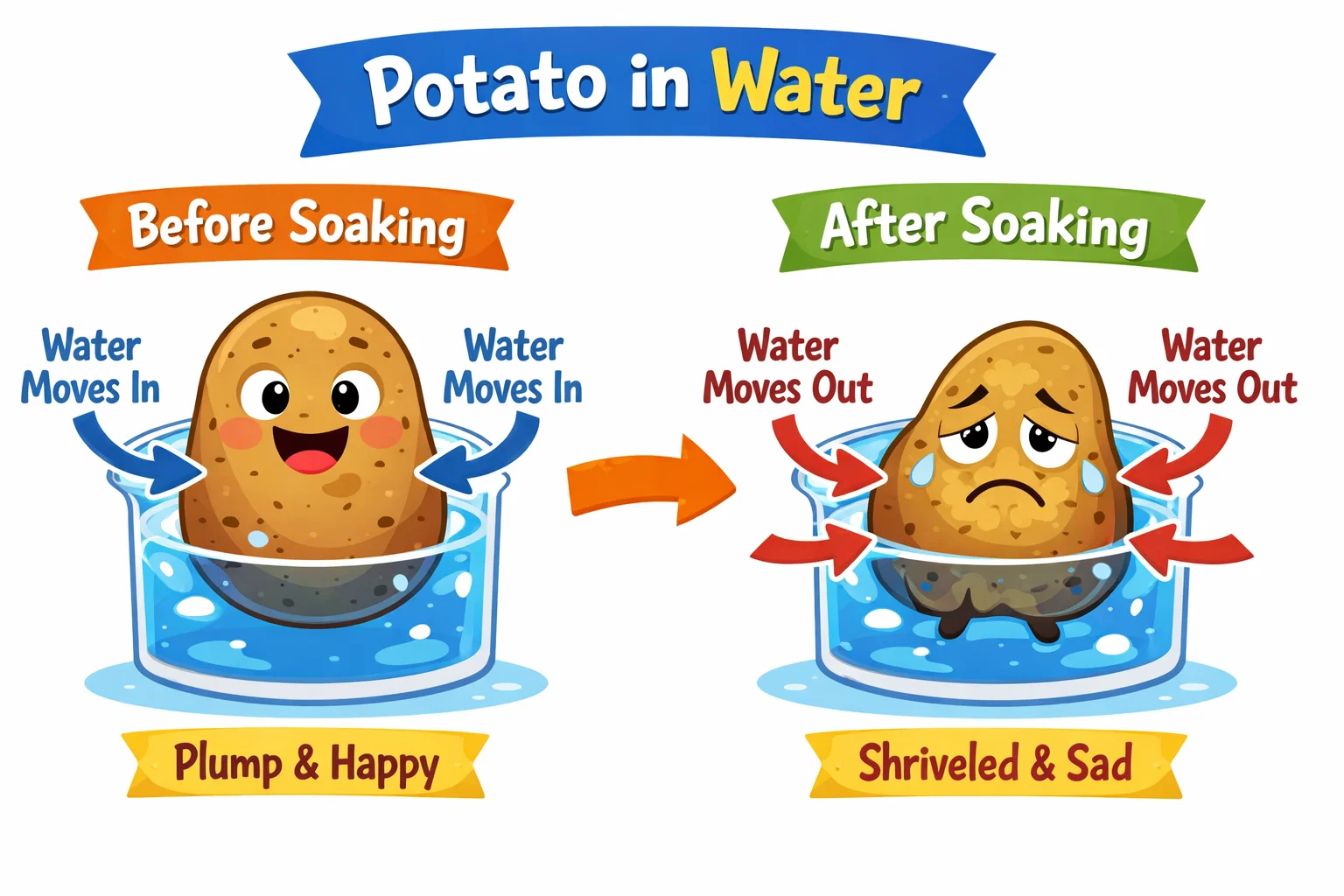
At its core, the potato osmosis experiment is a study of balance. It demonstrates how water molecules move in and out of plant cells to achieve equilibrium. Because a potato cell is surrounded by a membrane that acts like a tiny security gate, it only allows certain substances—like H₂O—to pass through.
In this experiment, kids will observe potato cylinders or slices placed in different solutions, such as pure water, a salt solution, or a sugar solution. By measuring the initial mass and the final mass, students can calculate the percentage change in mass, providing a clear “before and after” picture of osmosis in action.
Purpose of potato osmosis experiment
The primary goal is to visualize the movement of water across a semi-permeable membrane. While we can’t see individual molecules shifting, we can see the physical impact on the potato piece. The experiment aims to prove that water will move from an area of high water concentration to an area of lower water concentration to an area of low concentration (where there is less fluid). This helps children understand that cells are not static; they are constantly reacting to the environment around them.
Why potatoes work for osmosis experiments
These tubers are the “Goldilocks” of science specimens:
- Uniform Structure: They have a dense, consistent cellular makeup that makes it easy to cut identical potato strips.
- High Fluid Content: Since they are mostly moisture, the concentration inside the cells is easy to manipulate.
- Durability: Unlike delicate leaves, a potato slice holds its shape well enough to be handled, weighed, and measured by small hands.
- Turgor Pressure: You can physically feel the difference between a “stiff” specimen (full of liquid) and a “floppy” sample (lost moisture).
Age range and learning level suitability
This activity is incredibly versatile and can be adapted for various stages of development:
- Preschool/Early Primary: Focus on the “magic” of shrinking and growing. Use words like “thirsty” or “full.”
- Middle School: Introduce initial mass, final mass, and basic math to calculate weight differences.
- High School (GCSE Level): Focus on the movement of water by osmosis, hypotonic vs. hypertonic solutions, and creating a line of best fit on a results table.
Materials and Setup for Potato Osmosis Experiment
Setting up your potato osmosis lab is quick and budget-friendly. Most materials are already in your kitchen.
Materials list for potato osmosis experiment
- Tubers: Two or three large Russet or Yukon Gold specimens.
- Salt & Sugar: To create different concentrations.
- Liquid: Distilled water is best for a “pure” control, but regular water works too.
- Containers: Transparent cups or beakers (labeled).
- Tools: A knife (for adult use) or an apple corer to make potato cylinders.
- Measuring Gear: A digital kitchen scale (for mass) and a ruler (for length).
- Cleanup: Paper towels for gently drying samples before weighing.
Preparing potato pieces
Consistency is the key to a fair test. You should cut all samples into uniform shapes.
- Peel the tuber (the skin can block the movement of water).
- Slice the specimen into strips—aim for about 5cm long and 1cm wide.
- Ensure each potato piece looks identical. If one is significantly larger, it will skew your final mass results.
Preparing water, salt, and sugar solutions
To see a variety of results, you need different solutions. Label your cups clearly:
- Cup A (Control): Pure water (0.0 concentration).
- Cup B (Low Salt): 1 teaspoon of salt dissolved in 1 cup of water.
- Cup C (High Salt): 3 tablespoons of salt dissolved in 1 cup of liquid (higher salt concentration).
Advanced Option: For greater precision in school science projects for a science project, use molarity (e.g., 0.2M, 0.4M, 0.6M) to create a gradient of sucrose or salt. This allows for a more detailed math analysis.
Step-by-Step Potato Osmosis Experiment Instructions

Follow these steps to ensure your potato osmosis experiment yields clear, scientific data.
Initial measurements and observations
Before the samples go for a swim, you must record their starting state.
- Assign a number to each potato slice.
- Weigh each piece and record the initial mass in your results table.
- Note the texture (firm or flexible).
- Record the length in millimeters using a precise ruler.
Soaking potatoes in different solutions
- Put one potato piece into each labeled cup.
- Ensure the sample is completely submerged in the sugar solution or salt mixture.
- Leave the potato pieces for at least 30 minutes, though 2 to 24 hours provides much more dramatic results.
- Keep the cups in the same environment (same temperature) to keep the experiment fair.
Final measurements and comparison
- Remove the pieces of potato from their cups.
- Dry with a paper towel gently to remove excess surface moisture—this ensures you are only weighing the mass of the potato itself.
- Record the final mass.
- Compare the weights and calculate the percentage change in mass using this formula:
Percentage Change (%) = (Final Mass − Initial Mass) ÷ Initial Mass × 100
What Happens to Potatoes in Salt Water?
This is the “aha!” moment for kids. When you place a plant cell or tissue into a solution that has a higher salt concentration than its own cells, something specific happens.
Potato texture and size changes
In a salt solution, you will notice the sample becomes flexible, “floppy,” and smaller. It might feel like a piece of rubber rather than a crisp vegetable. Conversely, in distilled water, the specimen often becomes even firmer and may slightly increase in size.
Water movement explanation for kids
Imagine the water molecules are like kids at a party. They like to move where there is more “room” to dance. In the salty cup, there are a lot of salt molecules taking up space, so the water concentration is lower outside the tuber. The moisture inside the potato cell decides to move from an area of high concentration (inside the tissue) to an area of low concentration (the salty fluid) to try and balance things out. This process of moving water across the semi-permeable cell membrane makes the specimen lose weight.
Differences between plain water and salt water
| Feature | Plain Liquid (Hypotonic) | Salt Solution (Hypertonic) |
| Moisture Movement | Water moves into the sample | Water moves out of the sample |
| Mass Change | Increase in final mass | Decrease in final mass |
| Texture | Firm and Turgid | Soft and Plasmolyzed |
| Cell State | Turgid | Plasmolyzed |
Data Collection and Observation Ideas

Turning a fun activity into a science project requires good data. Quantitative analysis is a huge part of biology!
Measuring length, mass, and firmness
While mass is the most accurate way to compare the results, measuring length is a great backup. You can also create a “Firmness Scale” from 1 to 5 to track the potato osmosis experiment progress.
- 1: Very floppy (like cooked pasta)
- 5: Very stiff (snaps when bent)
Recording results in table or chart
Encourage kids to draw a results table. This keeps the data organized and makes it easier to make a graph later.
| Sample ID | Solution Type | Initial Mass (g) | Final Mass (g) | Change (g) | % Change |
| 1 | Pure Fluid | 10.2 | 11.5 | +1.3 | +12.7% |
| 2 | 5% Salt | 10.1 | 9.4 | -0.7 | -6.9% |
| 3 | 20% Salt | 10.3 | 8.2 | -2.1 | -20.4% |
Observing patterns and trends
Ask your child: “Does more salt mean more shrinking?” They should notice a trend: the higher salt concentration the solution has, the more the mass of the potato decreases. This often shows a clear trend that can be plotted on a graph with a line of best fit.
Osmosis Experiment Variations With Potatoes
Once you’ve mastered the basic potato osmosis lab, try these variations to deepen the learning.
Salt solutions of different concentrations
Instead of just “salt” and “no salt,” create a range: 0%, 1%, 2%, 5%, and 10%. This allows you to find the “isotonic point”—the exact concentration salt level where the mass of the potato doesn’t change at all because the concentration inside its cells matches the fluid outside.
Sugar solution osmosis experiment
Does sucrose (sugar) work the same as salt? Yes! Osmosis is the movement of water across a semi-permeable membrane regardless of the solute. However, because sugar molecules are larger than salt molecules, the rate of diffusion might vary. It’s a great way to compare the results between two different types of solutes.
Effects of temperature on osmosis
Water molecules move faster when they are warm. Try putting three potato pieces in cold fluid, room temperature liquid, and warm moisture (not boiling!). You’ll likely find that the movement of water happens much more quickly in the warm solution.
Science Concepts Behind Potato Osmosis
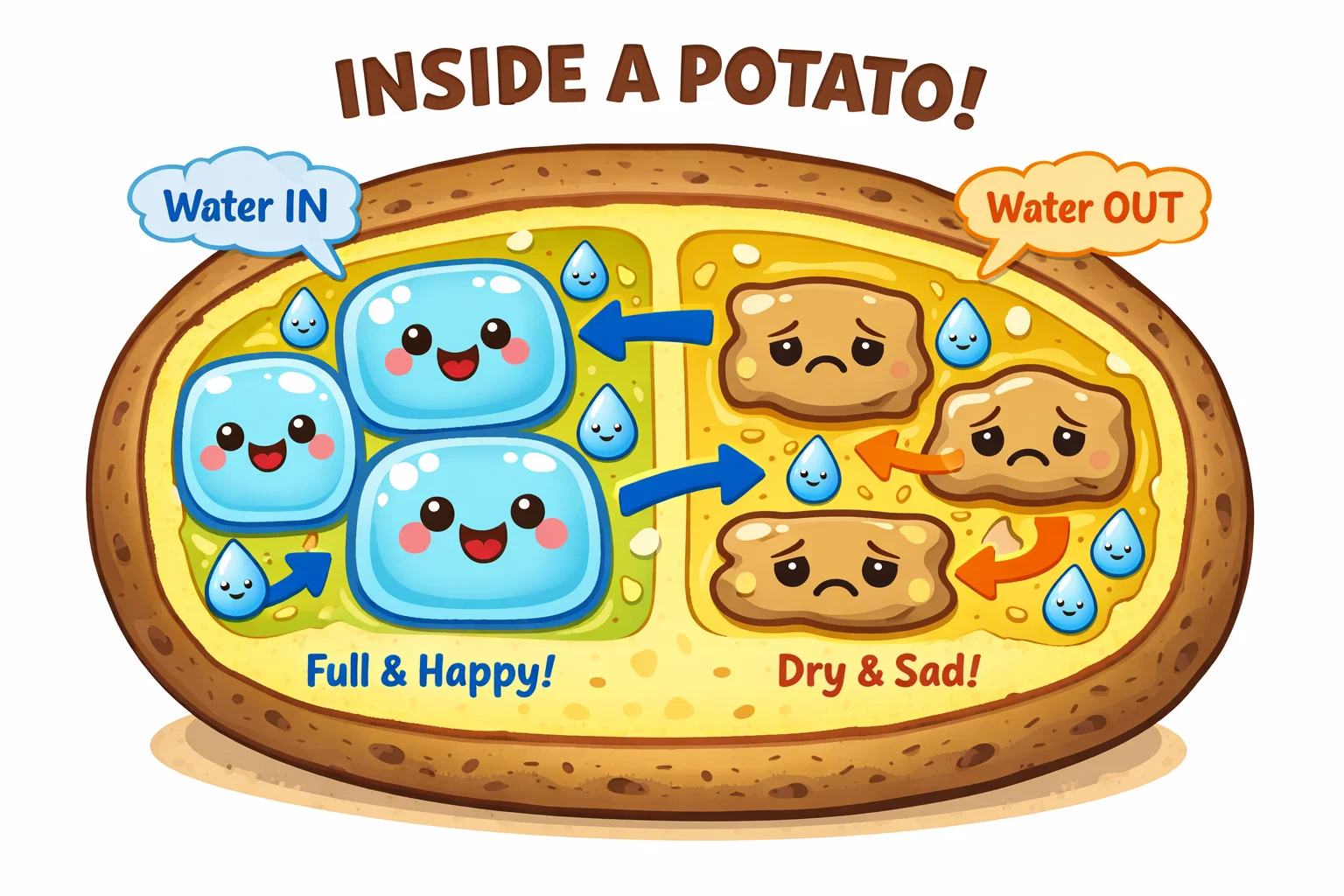
Let’s get into the “why.” Understanding the concept of osmosis is a gateway to biology.
Plant cells and water balance
Plant cells have a rigid cell wall. When they are full of moisture, they push against that wall, making the plant stand up straight. This is called turgor pressure. When you leave the potato in saltwater, the cells lose liquid and the pressure drops, which is why the potato slice becomes floppy.
Role of concentration differences
Osmosis is specifically the diffusion of water across a semi-permeable membrane.
- Hypotonic: The solution has less salt (more moisture) than the cell. H₂O enters the cell.
- Hypertonic: The solution has higher salt concentration (less moisture) than the cell. H₂O leaves the cell.
- Isotonic: The concentrations are equal. No net water movement.
Linking experiment to real plants
This isn’t just a potato lab; it’s how nature works. When you over-fertilize a garden, you accidentally create a higher salt concentration in the soil. This sucks the fluid out of the plant roots due to osmosis, causing the plants to wilt. On the flip side, roots use osmosis to pull pure water from the ground into the plant.
Classroom and Home Learning Applications
School science project ideas
This experiment is perfect for a science fair. To make it stand out:
- Use a large sample size (e.g., three potato pieces per cup) to find an average.
- Make a graph showing the relationship between sucrose concentration and percentage change in mass.
- Take time-lapse photos of the pieces of potato as they change over time.
STEM learning extensions
- Math: Calculate the surface-area-to-volume ratio of your potato cylinders and see if it affects the speed of osmosis.
- Chemistry: Discuss why salt (NaCl) dissociates in moisture and how that affects the concentration.
Safety tips for kids experiments
- Adult Supervision: An adult should always use a knife to cut the vegetables.
- Allergies: While rare, some people have skin sensitivities to raw starch.
- Disposal: The saltwater and sugar solution can be poured down the drain, and the scraps can be composted.
Similar Science Experiments for Kids
If your child loved the potato osmosis experiment, they might enjoy these related activities:
Other osmosis experiments for kids
- Naked Egg: Dissolve an eggshell in vinegar, then place the “naked” egg in syrup or fluid to see it shrink and grow.
- Gummy Bear Science: Gummy bears are great for showing water movement as they swell to massive sizes in regular water.
Plant and cell science experiments
- Celery Transpiration: Put celery stalks in food-colored fluid to see how plants transport moisture upwards through the xylem.
- Leaf Chromatography: Use rubbing alcohol to separate the colors (pigments) in a leaf.
Easy biology experiments at home
- Yeast Inflation: Use sugar and warm moisture to “wake up” yeast and blow up a balloon.
- Seed Germination: Grow beans in a plastic bag with a damp paper towel to observe root growth.


